 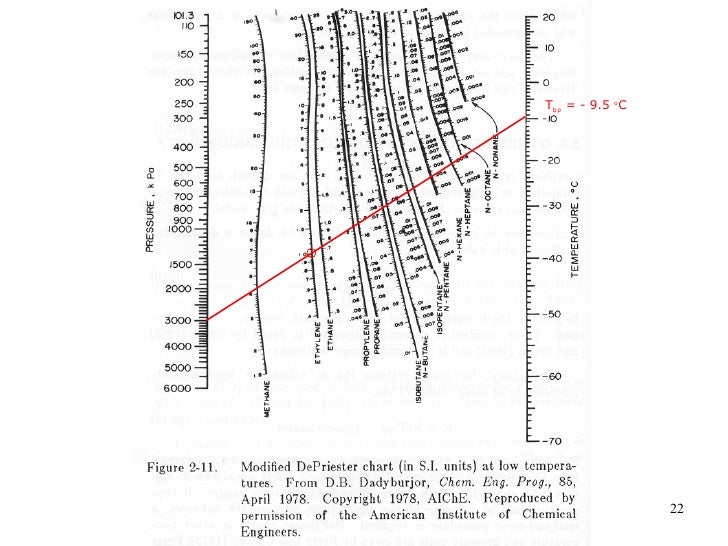
There is no single K-Value correlation that is superior for all mixtures encountered. 
The accuracy of the results of calculations involving K-Values depends on the reliability of sampling, of the analysis of that sample, and the K-Value correlation used. Therefore, the following guidelines extracted from page 128 of Vol 1 of JMC book are suggested. Again, the calculated liquid fractions by the Raoult’s law and Wilson correlations are close to each other but they deviate considerably from the GPA charts and the SRK EoS results.ĭue to the observation made in the previous section and other studies, care must be taken in selecting K-values correlations. 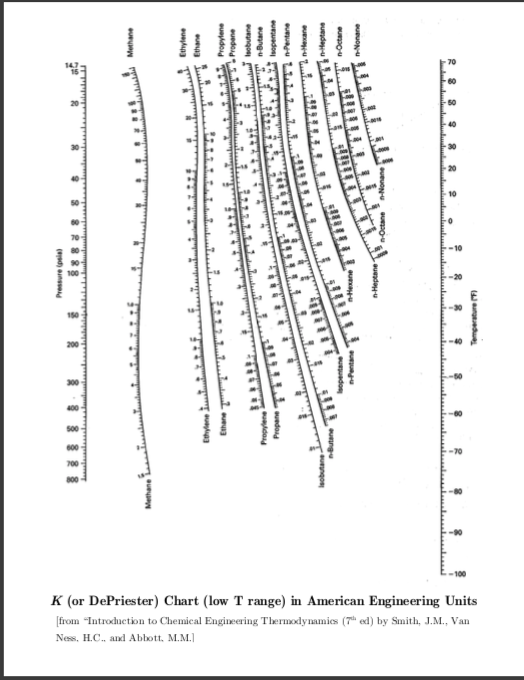
Similarly, for the same mixture shown in Table 1, a series of flash calculations for two isotherms were performed and the calculated liquid fractions (L/F) using different methods are compared in Table 5. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
